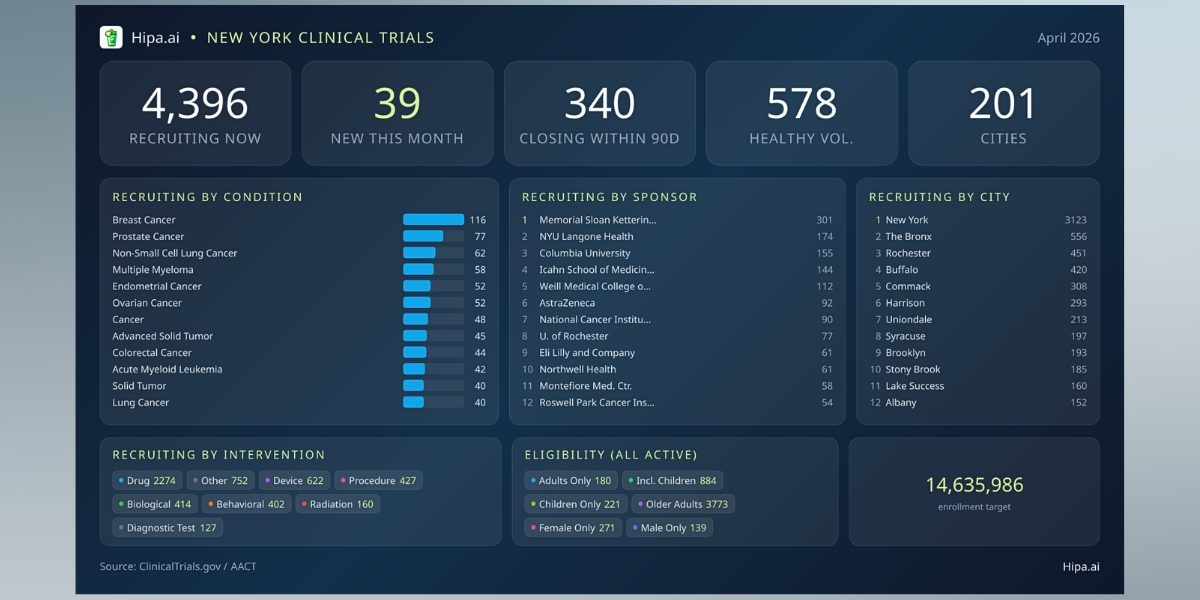
The most interesting clinical trial story in New York City this spring is not which new drugs are being tested. It is becoming much easier to find one of these studies and actually take part.
In January 2026, the Mount Sinai Tisch Cancer Center deployed an AI-driven matching platform called PRISM that connects patients across the entire Mount Sinai Health System to active cancer trials, including patients seen at the system’s Brooklyn, Queens, and Long Island sites who previously had to be transferred to Manhattan to be considered for a study. It was the first NCI-designated comprehensive cancer center in New York City to deploy oncology-specific AI for trial matching at this kind of scale. That single deployment is part of a broader pattern also visible in the latest research data for April 2026.
The state opened 39 new clinical trials in April. That is a slower month than March (70 new trials) and February (100), but the deeper story is in the design of those trials. Several were built specifically to lower the barriers that have historically kept New Yorkers from contributing to research: long commutes, multiple in-person visits, and narrow eligibility criteria.
If you want to see what is open near you right now, the Hipa.ai directory of clinical trials currently recruiting in New York City sorts active studies by condition, age, and healthy-volunteer eligibility, with breakouts for each borough.
When You Don’t Have to Leave Your Apartment to Be in a Study
The clearest example from April is the University of Rochester’s NCT07505162, a 1,000-person observational study of myotonic dystrophy that runs entirely from participants’ homes. Patients with myotonic dystrophy face muscle weakness, fatigue, and mobility challenges, exactly the kinds of issues that have historically excluded them from research that required regular clinic visits. A no-travel design opens the door for people who want to contribute but cannot regularly get to a research center.
Decentralized trial design is not new, but the volume is. According to CB Insights’ 2026 scouting report on AI in clinical development, more than 40% of the clinical research startups it tracks are now working on decentralized trials or real-world evidence collection. That shift is no longer just an industry conference talking point. It is appearing in the study protocols being filed at ClinicalTrials.gov, including the Rochester trial above.
For New Yorkers, the practical consequence is that the question “is there a trial open for my condition” no longer has to be paired with “and can I get there twice a week for six months.”
Where the Research Sits
The geographic concentration of New York’s clinical research is striking. Of the 39 new April trials, 26 launched in New York City itself, with smaller clusters on Long Island (Commack and Harrison with 5 each, Rockville Center and Uniondale with 2 each) and the outer boroughs (the Bronx 4, Brooklyn 3). Upstate sites added Rochester (3), Syracuse (2), and Dryden (1).
Across all 4,396 trials currently recruiting statewide, the bulk run at sites in or around the city. That density reflects the institutional cluster that defines New York medicine: Memorial Sloan Kettering Cancer Center, NYU Langone Health, Mount Sinai Health System, Weill Cornell Medicine, NewYork-Presbyterian, and Columbia University Irving Medical Center all run active research portfolios within a roughly fifteen-mile radius. For most New Yorkers, at least one of these institutions is reachable on a single subway or commuter rail trip.
Trials You Probably Don’t Hear About
A lot of what gets written about clinical trials in New York focuses on the headline oncology work happening at MSK and the Perlmutter Cancer Center at NYU Langone. April’s data shows a more diverse set of research underway.
Memorial Sloan Kettering itself is running a 750-participant Phase 3 trial (NCT07507942) testing whether a single dose of methylprednisolone administered before liver surgery reduces postoperative infections and shortens hospital stays. This is a pragmatic surgical question rather than a new-drug question, and a 750-person Phase 3 has the statistical weight to actually settle it.
Columbia University began a 500-participant project (NCT07517461) with a different goal entirely: reducing overuse of screening mammography in women aged 75 and older. The study does not test a treatment. It tests a multi-level intervention to change clinical practice patterns. It is one of the more interesting deimplementation studies open in the country right now and speaks to a less-visible side of clinical research: figuring out which existing practices should be scaled back.
On the metabolic side, Novo Nordisk launched parallel Phase 2 trials studying the same investigational compound in two different populations: 480 participants with both excess weight and Type 2 diabetes (NCT07531373, AMAZE 1) and 400 participants with excess weight and knee osteoarthritis (NCT07530937, AMAZE 4). Pairing one molecule with two distinct comorbidities is a way of asking whether the metabolic agent has effects that extend beyond glucose control.
How Compensation Actually Works
A practical note that often goes missing from coverage of clinical trials: most trials compensate participants for their time and travel, and the amount varies based on study design, time commitment, and the procedures involved. Healthy-volunteer studies tend to involve different structures than Phase 1 pharmacology trials, which require multiple overnight stays in a research unit. The exact compensation for any given trial is disclosed by the study team during the informed consent process.
Of the 39 new April trials, 11 are recruiting healthy volunteers, and 13 include older adults. Across all currently enrolling studies in New York, 578 are open to healthy volunteers. That is the broadest entry point for people without a specific diagnosis who want to contribute to medical research.
The Closing-Soon Window
For people who already have a diagnosis, the more time-sensitive number is on the other side of the ledger. 340 New York Medicaid plans are scheduled to stop accepting enrollees within the next 90 days. The largest closing-soon clusters by condition are breast cancer (12 trials), cancer (8), and glioblastoma (8), then a band of 4-trial groups including major depressive disorder, ovarian cancer, prostate cancer, pancreatic cancer, and Alzheimer’s disease. Mild cognitive impairment and lung cancer each have 3 trials reaching their close dates.
Trials often close enrollment earlier than the listed end date once recruitment quotas are met. A study listed as closing in 90 days can effectively close in weeks. For anyone who fits the eligibility criteria for one of these conditions, the time to actually apply is now, not later in the spring.
A More Compact City for Research
The story of April 2026 in New York’s clinical research is not really about volume. It is about how much more compact the on-ramp to participation has become. AI matching tools like Mount Sinai’s PRISM, remote-by-design protocols like the Rochester myotonic dystrophy study, and the broad institutional density of the city’s medical system have collectively turned what used to be a fragmented and somewhat opaque process into something a typical New Yorker can actually use. Hipa.ai publishes a refreshed New York report each month with updated condition splits, sponsor breakouts, and city-level trial counts.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Clinical trial participation involves potential risks and benefits that vary by study. Eligibility, procedures, and compensation are determined by each study’s sponsor and institutional review board, and full details are disclosed during the informed consent process. Readers considering enrollment should consult their healthcare provider and review the complete study protocol before making any decisions.